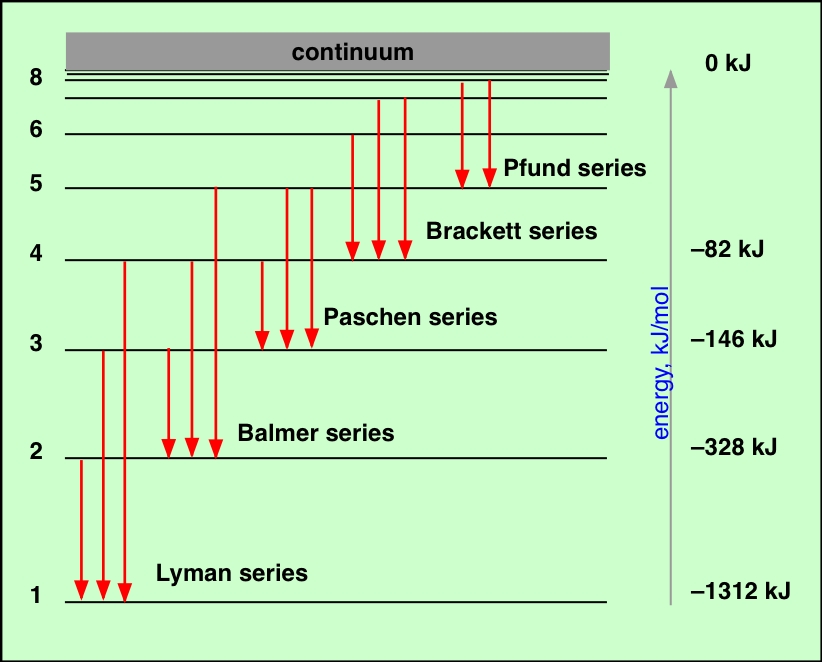
This means that line spectra can be used to identify elements.Ĭontinuous spectra are produced by electrons being shared between many atoms, giving a huge range of possible frequencies, as shown below. As a result each produces photons with different energy and so the line spectra for different elements will be different. This is not a continuous spectrum as only light of specific frequencies and specific colours are produced.ĭifferent types of atoms have different energy levels. This causes line emission spectra to be produced, as shown below. This means that each electron transition will produce a photon of a different frequency and hence a different colour. \(f\) is the frequency of light producedĪs the energy levels have different values, each of the possible electron transitions within an atom will produce a photon with a different energy.If an electron moves from level \(E_\) the energy of the photon can be worked out using the following: The energy of the photon can be worked out using the equation The amount of energy it loses will be equal to the difference in the energy levels it moves between. ISBN: 9780674834408.If an electron is in an excited state it can return to a lower energy level. Cambridge, MA: Harvard University Press, 1979. In an emission spectra electrons are excited to an excited state by thermal or electrical means and then relax back to a lower state and emit a photon of light at a specific energy, which is seen as at a specific wavelength. Cambridge, MA: Harvard University Press, 1963. There are two types of line spectra, emission and absorption. New York, NY: Cambridge University Press, 1996. Cecilia Payne-Gaposchkin: An Autobiography and Other Recollections. This lecture gives more details about the atomic spectra of hydrogen along with matter/energy interactions involving atomic hydrogen. Lecture Slides (PDF - 1.3MB) Lecture Summary Line spectra the Bohr model uses of emission and absorption spectra 6.3, “Atomic Spectra and Models of the Atom.” Describe the ground state of the gas phase atom.Īrchived Lecture Notes #1 (PDF), Sections 3, 5.This book discusses regularities in spectra emitted by monatomic gases. Explain trends in ionization energy across the periodic table. Atomic Spectra compiles papers on the highlighted developments in the atomic spectra.Calculate the energy of an electron in the ground state of hydrogen.Explain why the radius of the electron orbital takes multiple values that are discrete, quantized, and non-linear.Describe how the atomic spectra of hydrogen is produced.Learning ObjectivesĪfter completing this session, you should be able to: Sadoway discusses the shell model and quantum numbers ( Session 5). Energy levels of a particular atom or ion displayed in order of energy above the ground state. The light which atoms give off is made up of specific wavelengths, called lines observed by a spectroscope, the lines are, collectively, atomic spectra. Transition probabilities for the lines are also displayed where available. Session 3: Atomic Models: Rutherford
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
